VAERS is the Vaccine Adverse Event Reporting System for the United States. It is administered by General Dynamics for the U.S. Centers for Disease Control (CDC) and the U.S. Food and Drug Administration (FDA).
https://vaers.hhs.gov/
People and reporters who are unfamiliar with VAERS sometimes erroneously claim that VAERS reports are "self-reported" symptoms. Most reports are submitted by medical personnel and that can easily been seen by reading complex medical jargon in the free-form symtom text section. The reports related to death are clearly not self-reported. However, there are definately some reports submitted by the general public.
It is a U.S. federal crime to submit a fake report to VAERS. Even with the threat of federal prosecution, there will always be some law-breakers. People who would like to discredit VAERS would be the most likely to submit obviously false reports.
- It is one of the only tools where researchers, physicians and the general public can read and query all of the details of adverse reaction reports, including the notes as entered by independent medical personnel.
- It empowers individuals to read through full reports (not just biased summaries) and make their own decisions for themselves and their family.
- It allows physicians to read through case details to help them with recommendations for their patients.
- Most other countries provide biased summaries to the public. Sometimes, cases from physicians can be rejected by these governments or by private companies based on government agency guidance documents (Canada). Sometimes the governments sign lucrative deals with drug manufacturers (Canada, Israel).
- Drugs companies want to eliminate VAERS because the public has access to data outside of their public relations campaigns.
- Government bureaucrats who are part of the revolving door between drug companies and government agencies want to eliminate VAERS.
- Foreign governments who simply provide biased and incomplete summary information want to eliminate VAERS as it can be used to advocate for the general public.
- Politicians who accept drug company lobbying money want to eliminate VAERS.
- Certain social media companies that want to control the narrative for their drug company partners/advertisers want to eliminate VAERS.
- Media companies who constantly accept large amounts of advertising from drug companies want to eliminate VAERS and regularly publish biased "fact check" articles for the drug companies.
While VAERS isn't perfect, it is enormously useful and any attempt to eliminate it will be at the direction of drug companies and their corrupt government and private partners. If that happens, the data published by the government and media will be worthless (at best).
Yes, not all cases listed in VAERS are caused by COVID-19 vaccine drugs, even though most cases are submitted by medical personnel. Some of those reactions may be conincidental and some may be caused by the COVID-19 vaccine drugs. In order to get accurate idea of cases caused by COVID-19 vaccine drugs, please carefully read the next three FAQ answers below related to 1) this website's methodology of calculating case totals, 2) VAERS backlogged cases; and most importantly 3) the VAERS reporting rate. If you carefully read through the answers to the three questions below, you will know more about COVID-19 vaccine drug cases and VAERS than most.
The methodology used on this site total calculate total cases is as follows:
- Every Friday, this site downloads the raw VAERS data from VAERS in CSV file format.
- The number of cases found in the "2021VAERSDATA.CSV" file is linked by VAERS_ID to the same VAERS_ID in the "2021VAERSVAX.CSV" file that has a vaccination type of "COVID19".
- There are a small number of cases (approximately 0.1%) where the vaccination type is "UNK" (Unknown), but the symptom_text field in the "2021VAERSDATA.CSV" file indicates that it was a COVID-19 vaccine drug. On the June 11, 2021 download, this amounted to 472 of 326,709 total cases. These cases are added to the total.
- The totals for each symptom (death, blood clots, etc.) is calculated based on the table created from steps 1-3 above.
- This site only counts the deaths and adverse reactions listed in VAERS from the United States. As of June 4, 2021, there are 1,300 foreign deaths from COVID-19 vaccine drugs reported to VAERS that is not included in the total listed on this site.
There are some sites that may get their totals from an independent aggregator such as OpenVAERS and VaxPain.us. Others may use the VAERS interactive tool to get totals. Some may not query the symptom_text to find other cases (as in Step #3 above) and some site may have more sophisticated queries that pick up small numbers of cases that are missed here. Finally, some sites may inadvertently count more than COVID-19 vaccine drugs in their total.
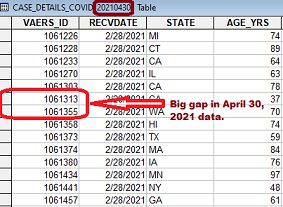
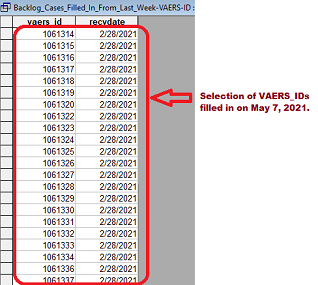
It is important to understand that each submitted case is given a sequential VAERS_ID number. In addition, there is a date field, RECVDATE (Received Date), to indicate the date it was submitted to VAERS. If you download several raw data files from 2021 and look at the csv file named "2021VAERSDATA.CSV" in each of the downloads, you will see many large gaps in the VAERS_ID numbers. The image on the left below shows a large VAERS_ID gap that could be seen in the April 30, 2021 data. The image on the right shows the gap filled in on May 7, 2021 for items received in February. Currently (June 11, 2021), there are 147,110 missing VAERS_ID's. These gaps are being filled by more than 10,000 cases every week that had already been received by VAERS weeks or months earlier. New gaps in higher VAERS_ID numbers are created each week that offset some of the gaps that are filled.
 |
 |
In order to see the gaps and how the gaps are filled every week, it is necessary to download several raw data files from 2021. There are other ways that investigators have seen gaps in data, such as by showing a huge decline in cases at some points of time when the distribution of COVID-19 vaccine drugs was increasing. (See this video, for example.)
What is just as important as a backlog of 147,110 missing cases in VAERS is that fact the CDC did not tell researchers, doctors and the general public that most of the submitted cases from 2021 have not yet been uploaded to VAERS! They presented the data as if it were up-to-date through June 4, 2021. The truth is only 69% of the submitted cases in 2021 have been made available.
Since February 1, 2021, approximately 0.4% of cases (4 in every 1,000 cases) are for non-COVID-19 vaccines. Starting on May 10, 2021 the calculations on this site estimate that 99.6% of the backlogged cases are related to COVID-19 vaccine drugs. (146,521 out of 147,110 backlogged cases in the June 4, 2021 VAERS data)
If you were to start a website to collect lung cancer cases from heavy tobacco use and then receive only five (5) cases in a one year timeframe, you might be tempted to proclaim that heavy tobacco use is fairly "safe," right? Wrong! You would know that those five cases represent only a small fraction of cases in the United States because the reporting rate to your website would be low. There may be countless thousands with tobacco-caused lung cancer who do not report to your website.
Similarly, the reporting rate to the VAERS website is key to estimating the number of total number of cases (reported + unreported) in the United States. There are four (4) pieces of evidence that point to a VAERS reporting rate of approximately 1%, meaning only 1 out of every 100 cases of symptoms or death linked to COVID-19 vaccine drugs are reported to VAERS:
- Harvard researchers found that less than 1% of adverse vaccine drug reactions are reported to VAERS. See the last two paragraphs of the Results section on page 6 of https://digital.ahrq.gov/sites/default/files/docs/publication/r18hs017045-lazarus-final-report-2011.pdf.
- Research looking at 64,900 persons who received the Pfizer or Moderna mRNA vaccine drug found the rate of serious anaphylaxis reactions was 50-100 times more than what was reported to VAERS. In other words, anaphylaxis reactions reported to VAERS represented only 1-2% of all anaphylaxis reactions after injection with the COVID-19 vaccine drugs.
- The U.S. Centers for Disease Control (CDC) presented in a slideshow on page 35 that they believe that only 1.13% of deaths expected at Long-Term Care Facilities (129 out of 11,440) were reported to VAERS after a COVID-19 vaccine drug injection.
- A former FDA Commissioner, presented a report
showing a 1% reporting rate for serious adverse drug reactions even when those reports are required.
The key point is that the reporting rate for VAERS adverse reactions are around 1% (1 in every 100 cases are reported). The reporting rate may be slightly higher for more severe reactions like death or anaphylaxis and slightly lower for less severe reactions.
Example: To estimate the number of cases of death in the U.S. linked to COVID-19 vaccine drugs, it would be calculated as follows:
- 4,618 deaths reported to and published by VAERS as of June 4, 2021.
- 2,071 estimated deaths cases reported to VAERS, but backlogged and not yet published as of June 4, 2021.
- 6,689 total cases (published by VAERS and backlogged) of death linked to COVID-19 vaccine drugs submitted to VAERS as of June 4, 2021.
- 668,900 Total Death Cases (Reported + Unreported) as of June 4, 2021 if we use a 1% reporting rate.
- 591,947 Total Death Cases (Reported + Unreported) as of June 4, 2021 if we use the CDC's estimate of 1.13% reporting rate.
This does not mean that all 668,900 (or 591,947) cases of death after an injection of a COVID-19 vaccine drug are caused by the drug. It is likely that some percentage of the cases are coincidental and some are caused by the drug. There are a couple of ways to get an idea of whether a significant percentage of cases are caused by the COVID-19 vaccine drugs:
- Compare the number of VAERS-reported cases of
death or a symptom linked to COVID-19 vaccine drugs to the number of cases of VAERS-reported symtoms or death from flu vaccines
(which were also injected well over a hundred millions times per year). If there is an unusual spike in reports only for COVID-19 vaccine drugs, then there is likely an
enormous problem.
- Look through the symptom text provided for each symptom on the data webpage (or query the VAERS database itself) to
see if many of the cases seem related to the COVID-19 vaccine drugs. A more sophisticated process would be for teams of independent experts (outside of the drug industry and
government) to look at the cases in more details. An appropriate team would be limited to researchers who have, prior to April 1, 2021,
publically called for more independent research and have no conflicts of interest. However, with 326,709 cases reported and 146,521 cases reported but backlogged
(as of June 11, 2021), a more sophisticated process could take years.